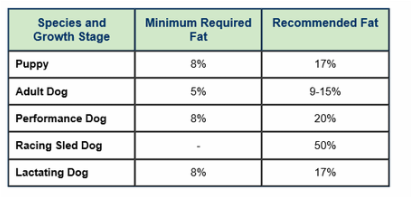
Figure 1: dog fat requirements(Foster and smith, 2015).
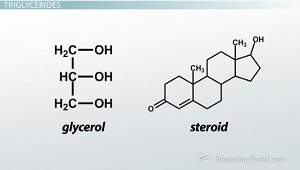
Figure 2: structure of fats
Structure
Fats consist of single molecules known as fatty acids. These are chains of hydrogen and carbon atoms that have a carboxyl group at an end and at the other end a methyl group. Carboxyl groups are made up of one carbon atom and one hydrogen atom and two oxygen atoms and methyl groups have one carbon and three hydrogen atoms. Carbon atoms that are in fatty acids molecules are connected by single or double bonds. (A fat molecule has 3 fatty acids connected to a glyceride). (Must have single bonds). Glycerol must have fat and a fatty acid needs a glycerol (Structure and function of lipids, 2015).
Fatty acids are different in size. Short fatty acid chain have two to four carbon atoms, medium chain have 6 to 12 carbon atoms, long have 14 to 18 carbon atoms. There are more than 20 carbon atom chains.
Fatty acids can be saturated or unsaturated. Saturated fatty acids have no double bonds between any carbon atoms in a chain. Unsaturated fatty acids have one or more double bonds in the carbon chain.
Monounsaturated fatty acids have one double bond and polyunsaturated fatty acids have no less than two double bonds. Unsaturated fatty acids are known by place of the double bond in the carbon chain.
Unsaturated fatty acids can have two different configurations of hydrogen atoms on each side of the double bonds. Cis configuration have hydrogen atoms both on the same side of the molecule. Making it look like the molecule is bent. Trans configurations have hydrogen atoms on different sides of the double bond. This shows a linear look similar to saturated fats.
Polypeptide chains; every amino acid and carboxyl group. A peptide bond; a dehydration reaction. It gets rid of water molecule, making a new bond. A polymer monomer is the new bond and dehydration makes them connect together.
A hydrolysis reaction puts water in a molecule; breaking the bond, this is hydrolysis of a polymer.
Lipids have a group of organic compounds these are: oils, fats and steroids. These are located in connected tissue and are made up of oxygen, carbon and hydrogen. They are broken into fatty acids and glycerol. There is no particular ratio of hydrogen to oxygen atoms. Every lipid is hydrophobic; does not break down in water.
Lipids are made up of two types of molecules 1 glycerol and 3 fatty acids. Triglycerides consist of 3 fatty acids and 1 glycerol. There is an Ester connection for a fatty acid and glycerol. Ester consists of carboxyl acid and alcohol. When it is heated under pressure of catalyst; sulphuric acid concentrated. Condensation reaction occurs; two molecules connect together to make a large molecule. (Ester and small molecule is water. An example of this is ethyl propanoate saturated for ease of break down.
Stearic acid; does not have double bonds and linoleic acid has double bonds this is because of the difference in melting point. In room temperature saturated fat molecules are squashed together to make a solid such as meats and animal fats. Unsaturated fat at room temperature is not able to stay together and so becomes a liquid form such as vegetable oil, sunflower oil and olive oil.
Hydrophilic heads and hydrophobic tails (phospholipid bilayer). Phospholipids is one of the fatty acids of triglyceride and is substituted with a phosphate group. Pieces are hydrophilic and some are hydrophobic.
Hydrophobic is scared of water and hydrophilic some substances do not dissolve.
Lipids cholesterol is a biological precursor of every steroid. (Testosterone, cholesterol, progesterone and oestrogen). Prostaglandin is an Arachidonic acid and they have: 20 carbon atoms in their fatty acid chains. An oxygen and hydrogen and carbon 11 and 15. When there is a Trans double bond the carbon is 13. It is created in injured tissues working pain, inflammation and fever. It is not made when anti-inflammatory drugs e.g. aspirin prevent their synthesis (Frayn, 2008).
Triglyceride chemical properties
Hydrogenation occurs when unsaturated compounds act with H2 C=C bonds________ C-C bonds
Hydrolysis occurs by been split by water and acid or an enzyme catalyst and creates glycerol and 3 fatty acids.
Hydrogenation swaps double bonds into oils which are single bonds. They are solid objects used to create margarine and they are divided into glycerol and 3 fatty acids (Frayn, 2008).
Function
A storage of energy for a long time. It constructs pieces of cells (phospholipid bilayer). Looks after vital organs by cushioning it, makes the diet taste better and gives absorption of fat soluble vitamins.
Fats become soft when heated and they have a sharp melting point. They cannot evaporate and by oxidation are broken down into a short chain (ketones and aldehydes). They do not smell or taste very nice when oxidized. They are a lubricant and cuts down friction in chewing and swallowing giving flavour to the foods.
When they are digested and absorbed the small intestine absorbs the lipids in the blood as lipoproteins. If they are not needed by the body they are kept as adipose tissue. It is synthesized from proteins and carbohydrates and used for energy from getting to cells. It is then goes to the brain and nervous tissue or it can be excreted in the faeces. It protects and supports vital organs, gives off heat for insulation and made back into a usable piece to be used for energy (Vance, 2008).
You need to put fat in the diet for feeding pregnant animals that are lactating because they require more energy to put weight on, they need a high fat, high calorie diet. Dogs and cats require a higher fat diet and chickens need more fat in their diet to make the food more palatable. Older animals need more cod liver oil in their diet to maintain joints and keep them lubricated.
Abnormal levels of fat can cause cardiovascular disease, artery disease, or even death in any animal.
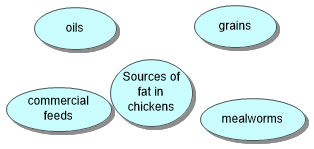
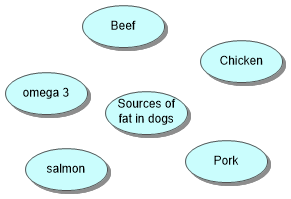
Reptiles get fats from the mealworms or crickets that they eat. Dogs get fat from meat in their commercial dog food. Cows get fat from grass and other vegetation. Fats are essential for carnivore reptiles needed for vitamin A, D and E.
Dairy cows that weigh 1,400lbs and produce 70lbs of milk a day will need 3.6% fat, 3.3% protein and 33Mcal a day of net energy for lactating. 0.2% linoleic acid is needed by reptiles however, it is not been proven to be essential in reptiles.
The digestibility of fat in dogs is 85-95 per cent as it is mixed with acylglycerols from plant and animal sources and fed. Commercial dry diets has 70-90 percent fat digestibility. The fat digestibility is not altered when vegetable protein was compared to beef in dogs. In younger dogs the digestibility of beef was dependent on unsaturated fatty acids concentration in the diet. Diets with 35 percent dry matter as fat and unsaturated acids lower than 40% of total dietary fat, the digestibility of beef reduces to 81-86 percent (Wiseman, 2013).
Fats consist of single molecules known as fatty acids. These are chains of hydrogen and carbon atoms that have a carboxyl group at an end and at the other end a methyl group. Carboxyl groups are made up of one carbon atom and one hydrogen atom and two oxygen atoms and methyl groups have one carbon and three hydrogen atoms. Carbon atoms that are in fatty acids molecules are connected by single or double bonds. (A fat molecule has 3 fatty acids connected to a glyceride). (Must have single bonds). Glycerol must have fat and a fatty acid needs a glycerol (Structure and function of lipids, 2015).
Fatty acids are different in size. Short fatty acid chain have two to four carbon atoms, medium chain have 6 to 12 carbon atoms, long have 14 to 18 carbon atoms. There are more than 20 carbon atom chains.
Fatty acids can be saturated or unsaturated. Saturated fatty acids have no double bonds between any carbon atoms in a chain. Unsaturated fatty acids have one or more double bonds in the carbon chain.
Monounsaturated fatty acids have one double bond and polyunsaturated fatty acids have no less than two double bonds. Unsaturated fatty acids are known by place of the double bond in the carbon chain.
Unsaturated fatty acids can have two different configurations of hydrogen atoms on each side of the double bonds. Cis configuration have hydrogen atoms both on the same side of the molecule. Making it look like the molecule is bent. Trans configurations have hydrogen atoms on different sides of the double bond. This shows a linear look similar to saturated fats.
Polypeptide chains; every amino acid and carboxyl group. A peptide bond; a dehydration reaction. It gets rid of water molecule, making a new bond. A polymer monomer is the new bond and dehydration makes them connect together.
A hydrolysis reaction puts water in a molecule; breaking the bond, this is hydrolysis of a polymer.
Lipids have a group of organic compounds these are: oils, fats and steroids. These are located in connected tissue and are made up of oxygen, carbon and hydrogen. They are broken into fatty acids and glycerol. There is no particular ratio of hydrogen to oxygen atoms. Every lipid is hydrophobic; does not break down in water.
Lipids are made up of two types of molecules 1 glycerol and 3 fatty acids. Triglycerides consist of 3 fatty acids and 1 glycerol. There is an Ester connection for a fatty acid and glycerol. Ester consists of carboxyl acid and alcohol. When it is heated under pressure of catalyst; sulphuric acid concentrated. Condensation reaction occurs; two molecules connect together to make a large molecule. (Ester and small molecule is water. An example of this is ethyl propanoate saturated for ease of break down.
Stearic acid; does not have double bonds and linoleic acid has double bonds this is because of the difference in melting point. In room temperature saturated fat molecules are squashed together to make a solid such as meats and animal fats. Unsaturated fat at room temperature is not able to stay together and so becomes a liquid form such as vegetable oil, sunflower oil and olive oil.
Hydrophilic heads and hydrophobic tails (phospholipid bilayer). Phospholipids is one of the fatty acids of triglyceride and is substituted with a phosphate group. Pieces are hydrophilic and some are hydrophobic.
Hydrophobic is scared of water and hydrophilic some substances do not dissolve.
Lipids cholesterol is a biological precursor of every steroid. (Testosterone, cholesterol, progesterone and oestrogen). Prostaglandin is an Arachidonic acid and they have: 20 carbon atoms in their fatty acid chains. An oxygen and hydrogen and carbon 11 and 15. When there is a Trans double bond the carbon is 13. It is created in injured tissues working pain, inflammation and fever. It is not made when anti-inflammatory drugs e.g. aspirin prevent their synthesis (Frayn, 2008).
Triglyceride chemical properties
Hydrogenation occurs when unsaturated compounds act with H2 C=C bonds________ C-C bonds
Hydrolysis occurs by been split by water and acid or an enzyme catalyst and creates glycerol and 3 fatty acids.
Hydrogenation swaps double bonds into oils which are single bonds. They are solid objects used to create margarine and they are divided into glycerol and 3 fatty acids (Frayn, 2008).
Function
A storage of energy for a long time. It constructs pieces of cells (phospholipid bilayer). Looks after vital organs by cushioning it, makes the diet taste better and gives absorption of fat soluble vitamins.
Fats become soft when heated and they have a sharp melting point. They cannot evaporate and by oxidation are broken down into a short chain (ketones and aldehydes). They do not smell or taste very nice when oxidized. They are a lubricant and cuts down friction in chewing and swallowing giving flavour to the foods.
When they are digested and absorbed the small intestine absorbs the lipids in the blood as lipoproteins. If they are not needed by the body they are kept as adipose tissue. It is synthesized from proteins and carbohydrates and used for energy from getting to cells. It is then goes to the brain and nervous tissue or it can be excreted in the faeces. It protects and supports vital organs, gives off heat for insulation and made back into a usable piece to be used for energy (Vance, 2008).
You need to put fat in the diet for feeding pregnant animals that are lactating because they require more energy to put weight on, they need a high fat, high calorie diet. Dogs and cats require a higher fat diet and chickens need more fat in their diet to make the food more palatable. Older animals need more cod liver oil in their diet to maintain joints and keep them lubricated.
Abnormal levels of fat can cause cardiovascular disease, artery disease, or even death in any animal.
Reptiles get fats from the mealworms or crickets that they eat. Dogs get fat from meat in their commercial dog food. Cows get fat from grass and other vegetation. Fats are essential for carnivore reptiles needed for vitamin A, D and E.
Dairy cows that weigh 1,400lbs and produce 70lbs of milk a day will need 3.6% fat, 3.3% protein and 33Mcal a day of net energy for lactating. 0.2% linoleic acid is needed by reptiles however, it is not been proven to be essential in reptiles.
The digestibility of fat in dogs is 85-95 per cent as it is mixed with acylglycerols from plant and animal sources and fed. Commercial dry diets has 70-90 percent fat digestibility. The fat digestibility is not altered when vegetable protein was compared to beef in dogs. In younger dogs the digestibility of beef was dependent on unsaturated fatty acids concentration in the diet. Diets with 35 percent dry matter as fat and unsaturated acids lower than 40% of total dietary fat, the digestibility of beef reduces to 81-86 percent (Wiseman, 2013).