Water
Water consists in 60-70% of the body and is required for all living cells in the body to function correctly. It allows materials to be moved from tissues to cells, controls pH balance, electrolyte balance, maintains body temperature, fights infection, and lubricates tissues. It also allows food to be pushed down the oesophagus (peristalsis) In the form of Saliva. Saliva consists of water and blood requires water so it does not clot and become hard. Water regulates body temperature and puts moisture into mucous membranes, lubricates joints (in the form of synovial fluid), aids digestion and stops constipation.
Water sources are: chemical reactions, drinking and eating.
It is lost through: sweat, urine, breathing, bodily fluids and faeces.
Within animals: water can be sourced through their normal diet. For example:
In dogs this can be from wet food and Lions get water from meat and blood.
Water and minerals
Minerals are mainly found in an animals normal diet.
Water also makes up part on Minerals. Minerals maintain body processes, provide structure to objects in the body, have no calories (energy), and are not able to be killed by heat so they are inorganic. They are not able to be synthesized from other things so they have to be given in their diet, However, the exception to this rule is Ash; this is left over as an inorganic material once combustion of organic matter such as minerals has occurred.
(Stone, 2009).
(Water Molecule structure, 2014).
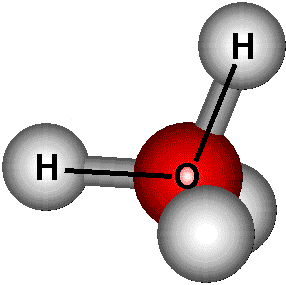
Chemical structure of water.
Water has two Hdrogen atoms connected to a single oxygen atom (H2O). The molecules of water are small and shaped like a V. The chemical formula for water is the H2O molecule. This molecule has 1 Oxygen atom and 2 Hydrogen atoms. At every Hydrogen nucleus is a bond to the central oxygen atom by a pair of electrons that are shared between them. At the Oxygen end of the bond is a negative charge needed by the slightly nonbonding electrons. This is an electric dipole.The Hydrogen electrons create a positive charge to bond the atoms together.H20 is surrounded by electrons formed in 2 layers (outer and inner). There are only 2 out of the 6 outer shell electrons from the original oxygen atom used, 4 electrons are prepared into 2 non-bonding pairs. There are four electron pairs covering the oxygen and organize themselves a fair distance away from one another to reduce repulsions between the negative charge. Due to the two non-bonding pairs staying close to the oxygen atom, they have stronger repulsions against the covalent bonding pairs, pushing the two hydrogen atoms nearer to each other.
The H2O molecule is electrically neutral; due to the negative and positive charges not being spread out evenly. (Elliot, 2014). This is due to Hydrogen needing the positive end of the bond. These bonds have a positive and a negative end due to the electrons not being shared equally. H2O molecules are squashed together in the liquid, the attractive forces have a hydrogen bonding. At low temperatures water freezes into ice where hydrogen bonds make a rigid and secure network. The hydrogen bond is longer than the covalent bond but it is a lot weaker (Water and hydrogen, 2009).
Water with animals.
All animals require a sufficient amount of water in their diet. This varies depending on the species, environment and lifestage.
For example:
A dog in the geriatric life stage will require less water then a puppy. This is because a puppy is more active and are still developing meaning they need a larger amount of water for their size to help their body function.
A chicken is really active and so needs water to keep hydrated. They also have grit in their diet, meaning they need more water to make the grit palatable and aid digestion.
The requirements for each animals water intake is:
Water consists in 60-70% of the body and is required for all living cells in the body to function correctly. It allows materials to be moved from tissues to cells, controls pH balance, electrolyte balance, maintains body temperature, fights infection, and lubricates tissues. It also allows food to be pushed down the oesophagus (peristalsis) In the form of Saliva. Saliva consists of water and blood requires water so it does not clot and become hard. Water regulates body temperature and puts moisture into mucous membranes, lubricates joints (in the form of synovial fluid), aids digestion and stops constipation.
Water sources are: chemical reactions, drinking and eating.
It is lost through: sweat, urine, breathing, bodily fluids and faeces.
Within animals: water can be sourced through their normal diet. For example:
In dogs this can be from wet food and Lions get water from meat and blood.
Water and minerals
Minerals are mainly found in an animals normal diet.
Water also makes up part on Minerals. Minerals maintain body processes, provide structure to objects in the body, have no calories (energy), and are not able to be killed by heat so they are inorganic. They are not able to be synthesized from other things so they have to be given in their diet, However, the exception to this rule is Ash; this is left over as an inorganic material once combustion of organic matter such as minerals has occurred.
(Stone, 2009).
(Water Molecule structure, 2014).
Chemical structure of water.
Water has two Hdrogen atoms connected to a single oxygen atom (H2O). The molecules of water are small and shaped like a V. The chemical formula for water is the H2O molecule. This molecule has 1 Oxygen atom and 2 Hydrogen atoms. At every Hydrogen nucleus is a bond to the central oxygen atom by a pair of electrons that are shared between them. At the Oxygen end of the bond is a negative charge needed by the slightly nonbonding electrons. This is an electric dipole.The Hydrogen electrons create a positive charge to bond the atoms together.H20 is surrounded by electrons formed in 2 layers (outer and inner). There are only 2 out of the 6 outer shell electrons from the original oxygen atom used, 4 electrons are prepared into 2 non-bonding pairs. There are four electron pairs covering the oxygen and organize themselves a fair distance away from one another to reduce repulsions between the negative charge. Due to the two non-bonding pairs staying close to the oxygen atom, they have stronger repulsions against the covalent bonding pairs, pushing the two hydrogen atoms nearer to each other.
The H2O molecule is electrically neutral; due to the negative and positive charges not being spread out evenly. (Elliot, 2014). This is due to Hydrogen needing the positive end of the bond. These bonds have a positive and a negative end due to the electrons not being shared equally. H2O molecules are squashed together in the liquid, the attractive forces have a hydrogen bonding. At low temperatures water freezes into ice where hydrogen bonds make a rigid and secure network. The hydrogen bond is longer than the covalent bond but it is a lot weaker (Water and hydrogen, 2009).
Water with animals.
All animals require a sufficient amount of water in their diet. This varies depending on the species, environment and lifestage.
For example:
A dog in the geriatric life stage will require less water then a puppy. This is because a puppy is more active and are still developing meaning they need a larger amount of water for their size to help their body function.
A chicken is really active and so needs water to keep hydrated. They also have grit in their diet, meaning they need more water to make the grit palatable and aid digestion.
The requirements for each animals water intake is:
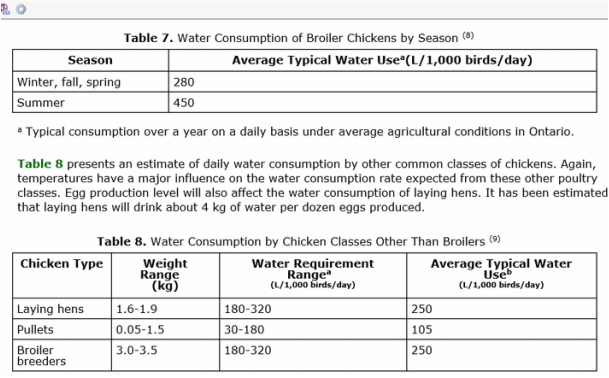
(Water requirements of livestock, 2014)
Dog requires 2.5 times more water a day than their food intake. Lions and all animals need water available at all times so they drink what they need.
