Protein
(Peptide, 2015).
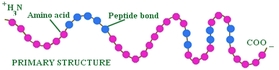
Figure 1: peptide bond
What is protein?
Proteins are in almost every cell. They also make up an organism, manage how it behaves and what it looks like. The body consists of thousands of proteins. Proteins use is to create fresh tissues for growth and repair. It is also used to control and maintain body functions. Enzymes are essential for immunity, protection and digestion. These all consist of proteins. Hormones require proteins for body regulation. Proteins can be used for energy when carbohydrates are not in the body. There is a high level of protein in the muscle tissue of all animals. Every cell breaks down proteins in their lifecycle. This is called protein synthesis. If this did not occur life could not be present. However this does not include animals that have intestinal micro flora and are able to synthesis protein from non protein sources.
Amino acids and proteins must be present in a diet to provide efficient growth and more functions in the body. Every cell is made up of protein and young animals need the most protein in their diet for growth. As animal ages they require less due to being less mobile and using less energy. When an animal is lactating and pregnant they require more protein in their diet because of can increased metabolic rate. This is due to carrying their offspring. When lactating, they also use more energy.
Ribosomes create the protein in the body. Protein is made up of carbon, hydrogen and nitrogen. Proteins transport materials such as blood. They are important for hair and nails (Ringe, 2004).
A protein strand has non polar side chains which are made up of mostly hydrocarbons. Amino acids are connected by peptide bonds to create protein whilst protein translation occurs. The amino acids are structured in a certain way due to DNA. DNA organises the amino acids into either primary, secondary or tertiary structures.
Primary structures
Primary proteins are a chain of amino acids. The primary structure of
proteins is the organisation that the amino acids connect to each otherto create a protein.
When you look at the whole protein chain to the left hand side you would see amino acid residue with an unconnected –NH2 group.
The N terminal is written on the left.
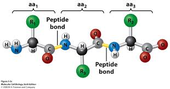
Combining amino acids
Peptide
When 2 amino acids are combined to create a dipeptide,
a molecule of water is eliminated. In the chemical structure,
the link joining the two amino acids is called a ‘peptide’ link
or ‘amide’ link. Once these amino acids have combined,
the chain no longer consists of amino acids due to the chemical
structure changing because of removing a molecule of water.
The peptide chain consists of the left over molecules of amino
acid substances.This is used for completing a wide range
of functions in the body.
If you draw a peptide chain the –NH2 group that has not been
converted into a peptide link is written at the left hand side.
The unaltered –COOH group is written at the right hand side.
The end of the peptide chain –NH2 is called the N terminal,
and the end –COOH group is called the C terminal.
An example of this is:
Protein use covalent bonds for creation. This is because every
peptide link consists of covalent bonds. When two cysteine
side chains are located next to each other due to the folding
of the peptide chain, they react to create a sulphur bridge.
This is a covalent link. The sulphur bridges depend on
how the protein folds; this is present in the tertiary structure below.
Tripeptide
When three amino acids are connected it is called a Tripeptide.
This is created by eliminating two molecules of water.
These are used to complete multiple functions in the body.
Polypeptide
If you joined a number of amino acids together in a chain it is
known as a polypeptide. A protein chain will consist of 50 to
2000 amino acid substances. These are created to complete many
functions in the body. These can work as a set chain,
broken into smaller chains or by combining the chains together.
The R chain determines the type of amino acid.
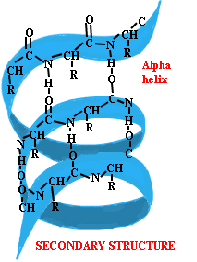
Secondary structures
Secondary structures of proteins are a local folding pattern of a polypeptide
backbone and stabilized by hydrogen bonds of N+H+C=O groups.
There are a number of different types of secondary structure the most
popular being orderly repeating structures called b- pleated sheet and helix.
In the long protein chains there are places where the chains are prepared
into a regular structure called alphahelices and beta pleated sheets.
The secondary structures are kept together by hydrogen bonds.
In one long pair there is always an oxygen atom and hydrogen and
nitrogen atoms are always connected.
The alpha helix, which is a protein chain, is coiled similar to a spring.
This coiling occurs in a clockwise manor as it comes away from you.
The alpa hydrogen bonds. When you have the full number of amino acid
residues at every turn, every group will have exactly the same group
underneath it. Hydrogen bonding is not capable when this happens.
Every turn has 3 complete amino acids residue and two atoms from the
following one. Every turn is offset from ones below and above,
e.g. N-H and C=O groups.
With Beta pleated sheets, the chains are folded lying alongside one another
. They can be antiparallel sheets, where the two chains go in opposite
directions. The folded chains are kept together by hydrogen bonds using
the same group as they do in the alpha helix (Hoove, 2010).
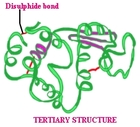
Tertiary structures
These are organised as a 3D polypeptide chain (helices, b sheets, folds and loops). These are formed when interactions take place between side chains and polypeptide backbone. These are formed when interactions take place between the side chains and the polypeptide backbone. Every tertiary protein has a specific pattern of folding and they can be very complex (Peet, 2012).
The whole chain folds into its 3 dimensional shape and this is simplified into models (enzyme dihydrofolate reductase). These enzymes are determined by amino acids. The alpha helices are coils of ribbon. The beta pleated sheets are flat parts of the ribbon ending as an arrow. The piece of the protein chain is randomly coiled and the loops are a similar structure to string.
The tertiary structure is kept together by interactions with side chains (R group). With ionic interactions particular amino acids are made up of an extra –COOH group. In addition particular amino acids consist of an extra –NH2 group. Hydrogen ions can be transferred from the –COOH to –NH2 group to make zwitterions the same as in simple amino acids. Ionic bonding can occur between negative and positive groups when the chain folds in such a way that they are near each other.
Quaternary structure
This is where the proteins are created by interactions between multiple polypeptide chains. Every polypeptide chain is a subunit. They contain multiple amounts of the same type of protein subunit. It is important because haemoglobin, DNA polymerase and ion channels are all essential for the body to work correctly and these are good examples of quaternary structure.
Different types of proteins
Collagen
Collagen is made up of tissue protein and is made up of a triple helix organised in parallel and quarter positioned to present the characteristic banded look of collagen. It is the most common structural protein in a number of tissues.
Elastin
Elastin is another protein tissue and looks similar to a denatured collagen made up of a long organised polypeptide chain. It is similar to rubber and is always found with a number of collagens in artery walls and ligaments.
Microfibrilar proteins (sarcoplasm)
Microfibrilar proteins are in homogenised muscle with dilute salts. They are made up of 20 enzymes that work with muscle metabolism.
Contractile proteins
These are myosin, actin and tropmyosin B; these are used in muscle contraction. Myosin is important in striated muscle as it is the thick filament. Keratins are present in hooves, beaks, hair, feathers, horns, claws and wool.
Blood proteins
They are also referred to as globulins. There are 3 types: gamma, beta and alpha. These are needed in the body to clean the blood.
The different types of protein that an animal can take into their body
Incomplete protein which is vegetable and complete which is animal protein. It is broken down, taken in and absorbed into the bloodstream and rebuilds again. In the stomach and small intestine protein is digested it is then taken in by the villi which cover the intestinal wall.
Protein synthesis
In order for protein synthesis to occur in animals nucleic acids need to be there as well. DNA transports genetic information in the cell and sends the signal of inherited characteristics from one generation to another. It is the outline to protein synthesis. DNA maintains the growing of a cell and organism by maintaining the creation of ribonucleic acid (RNA). These are all essential in protein synthesis.
Ribosomal RNA is a piece of the structure of the ribosome. This is the place where the creation of proteins occurs in the cell.
Transfer RNA transports particular amino acids to the ribosome, where they communicate with the messenger RNA. The messenger RNA decides the sequence of the amino acid in the protein being created.
Synthesis of every protein is maintained by a different messenger RNA. The information provided in protein synthesis is required in sufficient growth and development of animals.
The information is transferred by DNA to the place of protein synthesis in the cytoplasm is done by the RNA. Protein synthesis happens by transfer of amino acids to ribosomes. Particles are connected to membrane surfaces and amino acids which are connected in sequence by the sequence of nitrogenous bases in DNA and RNA. These ribosomes are important because they are basis of amino acid connections during protein synthesis. When more amino acids are added to the polypeptide chain it develops very quickly. Proteins connect themselves to receptor molecules on cell membranes. Improved muscle, lactating, and fetal growth have being because of protein in animals (Pond, 2005).
When proteins complex with carbohydrates they create glycoproteins. This comes from amino acids residues accepting sugars present in the polypeptide chain.
Essential amino acids are leucine, isoleucine, lysine, phenylalanine, methionine, tryptophan, threonine, valine and histidine. All of the other amino acids are able to be synthesized by the body.
Every protein will have a different sequence of amino acid. The different sequences identify different amino acids. These sequences are residues making up proteins.
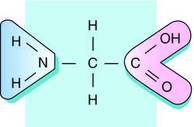
Amino acid is made up of the carboxyl group and the amino group bonding on either side of a C atom. A hydrogen atom is above the C atom. On a basic amino acid structure, an R side chain bonds below the C atom (Peet, 2012).
In acidic when the pH is less than 4 the –COO groups connect with the hydrogen ions and is converted into –COOH . In alkaline with pH above 9 the ammonium group gets rid of a hydrogen ion and changes it into an amino group (Mcneil, 2012).
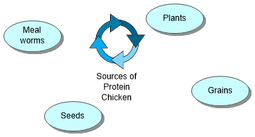
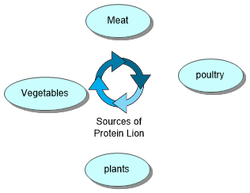
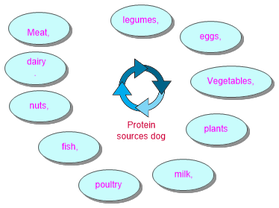
Proteins in different animals
Protein affects three different types of animals: Companion, production and collection animals.
All animals require more protein during pregnancy and when they are young as the animals need protein to grow and elderly animals need it to keep their bones strong.
Proteins are soluble in water insoluble in in organic solvents and they melt in high temperatures often 200 degrees C.
Proteins are in almost every cell. They also make up an organism, manage how it behaves and what it looks like. The body consists of thousands of proteins. Proteins use is to create fresh tissues for growth and repair. It is also used to control and maintain body functions. Enzymes are essential for immunity, protection and digestion. These all consist of proteins. Hormones require proteins for body regulation. Proteins can be used for energy when carbohydrates are not in the body. There is a high level of protein in the muscle tissue of all animals. Every cell breaks down proteins in their lifecycle. This is called protein synthesis. If this did not occur life could not be present. However this does not include animals that have intestinal micro flora and are able to synthesis protein from non protein sources.
Amino acids and proteins must be present in a diet to provide efficient growth and more functions in the body. Every cell is made up of protein and young animals need the most protein in their diet for growth. As animal ages they require less due to being less mobile and using less energy. When an animal is lactating and pregnant they require more protein in their diet because of can increased metabolic rate. This is due to carrying their offspring. When lactating, they also use more energy.
Ribosomes create the protein in the body. Protein is made up of carbon, hydrogen and nitrogen. Proteins transport materials such as blood. They are important for hair and nails (Ringe, 2004).
A protein strand has non polar side chains which are made up of mostly hydrocarbons. Amino acids are connected by peptide bonds to create protein whilst protein translation occurs. The amino acids are structured in a certain way due to DNA. DNA organises the amino acids into either primary, secondary or tertiary structures.
Primary structures
Primary proteins are a chain of amino acids. The primary structure of
proteins is the organisation that the amino acids connect to each otherto create a protein.
When you look at the whole protein chain to the left hand side you would see amino acid residue with an unconnected –NH2 group.
The N terminal is written on the left.
Combining amino acids
Peptide
When 2 amino acids are combined to create a dipeptide,
a molecule of water is eliminated. In the chemical structure,
the link joining the two amino acids is called a ‘peptide’ link
or ‘amide’ link. Once these amino acids have combined,
the chain no longer consists of amino acids due to the chemical
structure changing because of removing a molecule of water.
The peptide chain consists of the left over molecules of amino
acid substances.This is used for completing a wide range
of functions in the body.
If you draw a peptide chain the –NH2 group that has not been
converted into a peptide link is written at the left hand side.
The unaltered –COOH group is written at the right hand side.
The end of the peptide chain –NH2 is called the N terminal,
and the end –COOH group is called the C terminal.
An example of this is:
Protein use covalent bonds for creation. This is because every
peptide link consists of covalent bonds. When two cysteine
side chains are located next to each other due to the folding
of the peptide chain, they react to create a sulphur bridge.
This is a covalent link. The sulphur bridges depend on
how the protein folds; this is present in the tertiary structure below.
Tripeptide
When three amino acids are connected it is called a Tripeptide.
This is created by eliminating two molecules of water.
These are used to complete multiple functions in the body.
Polypeptide
If you joined a number of amino acids together in a chain it is
known as a polypeptide. A protein chain will consist of 50 to
2000 amino acid substances. These are created to complete many
functions in the body. These can work as a set chain,
broken into smaller chains or by combining the chains together.
The R chain determines the type of amino acid.
Secondary structures
Secondary structures of proteins are a local folding pattern of a polypeptide
backbone and stabilized by hydrogen bonds of N+H+C=O groups.
There are a number of different types of secondary structure the most
popular being orderly repeating structures called b- pleated sheet and helix.
In the long protein chains there are places where the chains are prepared
into a regular structure called alphahelices and beta pleated sheets.
The secondary structures are kept together by hydrogen bonds.
In one long pair there is always an oxygen atom and hydrogen and
nitrogen atoms are always connected.
The alpha helix, which is a protein chain, is coiled similar to a spring.
This coiling occurs in a clockwise manor as it comes away from you.
The alpa hydrogen bonds. When you have the full number of amino acid
residues at every turn, every group will have exactly the same group
underneath it. Hydrogen bonding is not capable when this happens.
Every turn has 3 complete amino acids residue and two atoms from the
following one. Every turn is offset from ones below and above,
e.g. N-H and C=O groups.
With Beta pleated sheets, the chains are folded lying alongside one another
. They can be antiparallel sheets, where the two chains go in opposite
directions. The folded chains are kept together by hydrogen bonds using
the same group as they do in the alpha helix (Hoove, 2010).
Tertiary structures
These are organised as a 3D polypeptide chain (helices, b sheets, folds and loops). These are formed when interactions take place between side chains and polypeptide backbone. These are formed when interactions take place between the side chains and the polypeptide backbone. Every tertiary protein has a specific pattern of folding and they can be very complex (Peet, 2012).
The whole chain folds into its 3 dimensional shape and this is simplified into models (enzyme dihydrofolate reductase). These enzymes are determined by amino acids. The alpha helices are coils of ribbon. The beta pleated sheets are flat parts of the ribbon ending as an arrow. The piece of the protein chain is randomly coiled and the loops are a similar structure to string.
The tertiary structure is kept together by interactions with side chains (R group). With ionic interactions particular amino acids are made up of an extra –COOH group. In addition particular amino acids consist of an extra –NH2 group. Hydrogen ions can be transferred from the –COOH to –NH2 group to make zwitterions the same as in simple amino acids. Ionic bonding can occur between negative and positive groups when the chain folds in such a way that they are near each other.
Quaternary structure
This is where the proteins are created by interactions between multiple polypeptide chains. Every polypeptide chain is a subunit. They contain multiple amounts of the same type of protein subunit. It is important because haemoglobin, DNA polymerase and ion channels are all essential for the body to work correctly and these are good examples of quaternary structure.
Different types of proteins
Collagen
Collagen is made up of tissue protein and is made up of a triple helix organised in parallel and quarter positioned to present the characteristic banded look of collagen. It is the most common structural protein in a number of tissues.
Elastin
Elastin is another protein tissue and looks similar to a denatured collagen made up of a long organised polypeptide chain. It is similar to rubber and is always found with a number of collagens in artery walls and ligaments.
Microfibrilar proteins (sarcoplasm)
Microfibrilar proteins are in homogenised muscle with dilute salts. They are made up of 20 enzymes that work with muscle metabolism.
Contractile proteins
These are myosin, actin and tropmyosin B; these are used in muscle contraction. Myosin is important in striated muscle as it is the thick filament. Keratins are present in hooves, beaks, hair, feathers, horns, claws and wool.
Blood proteins
They are also referred to as globulins. There are 3 types: gamma, beta and alpha. These are needed in the body to clean the blood.
The different types of protein that an animal can take into their body
Incomplete protein which is vegetable and complete which is animal protein. It is broken down, taken in and absorbed into the bloodstream and rebuilds again. In the stomach and small intestine protein is digested it is then taken in by the villi which cover the intestinal wall.
Protein synthesis
In order for protein synthesis to occur in animals nucleic acids need to be there as well. DNA transports genetic information in the cell and sends the signal of inherited characteristics from one generation to another. It is the outline to protein synthesis. DNA maintains the growing of a cell and organism by maintaining the creation of ribonucleic acid (RNA). These are all essential in protein synthesis.
Ribosomal RNA is a piece of the structure of the ribosome. This is the place where the creation of proteins occurs in the cell.
Transfer RNA transports particular amino acids to the ribosome, where they communicate with the messenger RNA. The messenger RNA decides the sequence of the amino acid in the protein being created.
Synthesis of every protein is maintained by a different messenger RNA. The information provided in protein synthesis is required in sufficient growth and development of animals.
The information is transferred by DNA to the place of protein synthesis in the cytoplasm is done by the RNA. Protein synthesis happens by transfer of amino acids to ribosomes. Particles are connected to membrane surfaces and amino acids which are connected in sequence by the sequence of nitrogenous bases in DNA and RNA. These ribosomes are important because they are basis of amino acid connections during protein synthesis. When more amino acids are added to the polypeptide chain it develops very quickly. Proteins connect themselves to receptor molecules on cell membranes. Improved muscle, lactating, and fetal growth have being because of protein in animals (Pond, 2005).
When proteins complex with carbohydrates they create glycoproteins. This comes from amino acids residues accepting sugars present in the polypeptide chain.
Essential amino acids are leucine, isoleucine, lysine, phenylalanine, methionine, tryptophan, threonine, valine and histidine. All of the other amino acids are able to be synthesized by the body.
Every protein will have a different sequence of amino acid. The different sequences identify different amino acids. These sequences are residues making up proteins.
Amino acid is made up of the carboxyl group and the amino group bonding on either side of a C atom. A hydrogen atom is above the C atom. On a basic amino acid structure, an R side chain bonds below the C atom (Peet, 2012).
In acidic when the pH is less than 4 the –COO groups connect with the hydrogen ions and is converted into –COOH . In alkaline with pH above 9 the ammonium group gets rid of a hydrogen ion and changes it into an amino group (Mcneil, 2012).
Proteins in different animals
Protein affects three different types of animals: Companion, production and collection animals.
All animals require more protein during pregnancy and when they are young as the animals need protein to grow and elderly animals need it to keep their bones strong.
Proteins are soluble in water insoluble in in organic solvents and they melt in high temperatures often 200 degrees C.
Figure 3: Secondary (Structure of proteins, 2014)
|
Figure 2: Glycine (smallest amino acid)
|
Figure 4: Tertiary (Proteins, 2014).
|